
We encourage you to take more examples and try determining the geometries yourself to grasp the concept further. 
We hope you now clearly understand how electron geometry and molecular geometry are different, and how they can help us understand the world of molecules and atoms better. The electron-pair geometries will be the same as the molecular structures when there are no lone electron pairs around the central atom, but they will be different when there are lone pairs present on the central atom. In such a way we can draw the structure of different molecules quite easily. Thus, the molecular geometry here is bent. So there are 4 electron groups and 2 lone electron pairs. Hydrogen donates a total of 2 electrons, making the total 8. Oxygen is the central atom here with 6 valence electrons. That’s how we determine that the electron geometry of CH4 is tetrahedral. The single bonds, in this case, are 4 and the number of lone pairs is 0. 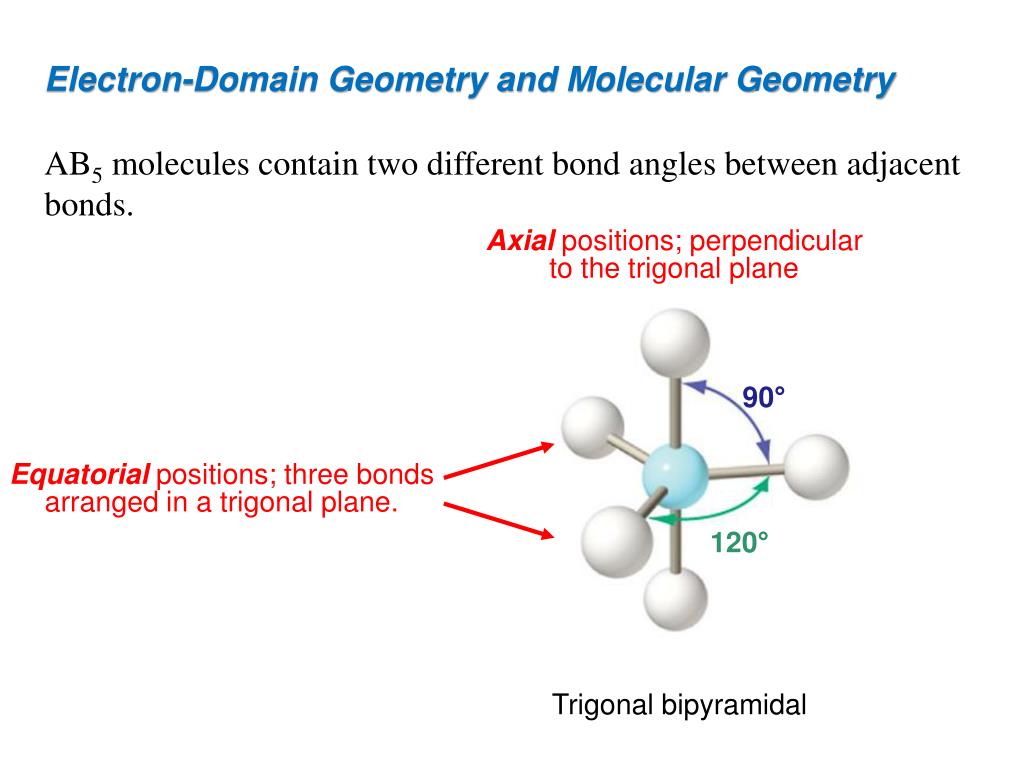
Hydrogen atoms donate 4 electrons, which means there are a total of 8 electrons around C. The central atom here is C, and there are 4 valence electrons. We can determine it through VSEPR theory, according to which, electron domains repel each other. Through electron geometry, we get the spatial arrangement of the lone pairs and bond in the molecule. Explore the relationship between the octet rule, valence electron, and the electron dot diagram. We calculate the number of total electron pairs in electron geometry and not in molecular geometry.ģ. 70K Learn to depict molecules and compounds using the Lewis structure. In molecular geometry, though, we only consider bond electron pairs.Ģ. We consider both lone electron pairs and bond electron pairs while determining the shape of a molecule in electron geometry. Electron Geometry vs Molecular Geometryġ. We use the electron domain geometry to help us predict the molecular geometry. To determine the shape of a molecule we distinguish between lone pairs and bonding pairs. The hydrogen atoms are as far apart as possible if opposite each other at 180 o. SEE SLIDES The molecular geometry is the arrangement of the atoms in space. This molecule is electron deficient and does not follow the octet rule because it has only 4 valence electrons. So, when you compare them, you will note that atoms have different arrangements in electron geometry and molecular geometry. Beryllium Hydride: Chime in new window An example of linear electron pair and molecular geometry is BeH 2. It is the 3D arrangement of all the atoms in a particular molecule. Electron domains for nonbonding electron. The effect of nonbonding electrons and multiple bonds on bond angles. Molecular geometry, on the other hand, helps us understand the entire atom and its arrangement. AJR Ch10 Molecular Geometry.docx Slide 10. To help you out, we are shedding light on electron geometry vs molecular geometry in this article.Įlectron geometry teaches us about the arrangement of different electron groups. However, a few concepts can be slightly difficult to comprehend because they seem similar or because they are just confusing! One such concept is the difference between electron geometry and molecular geometry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
